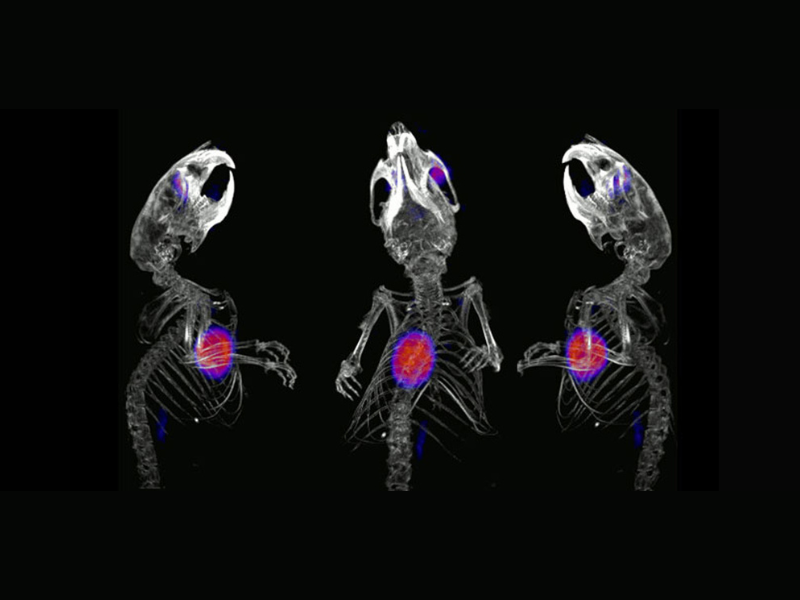
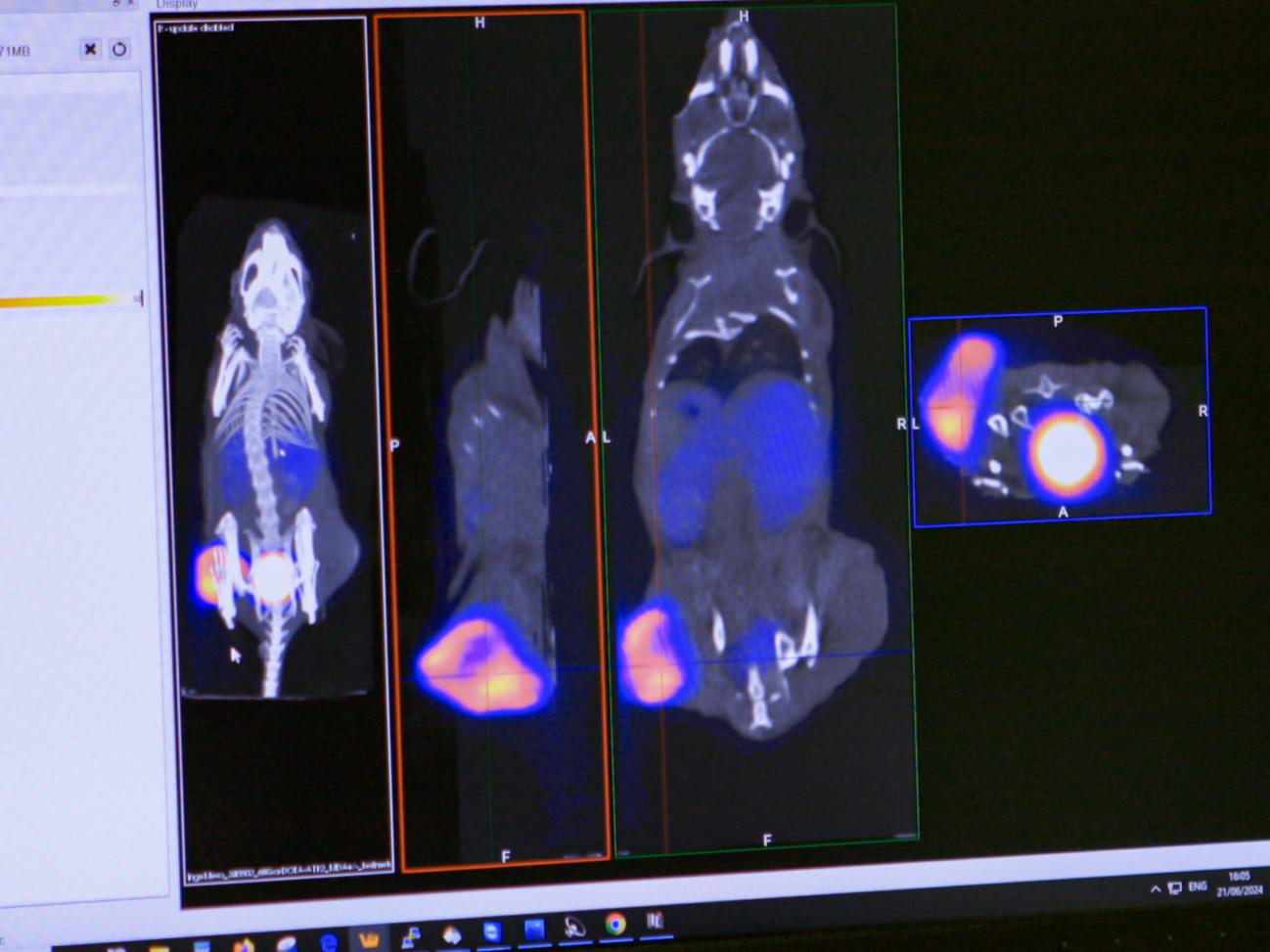
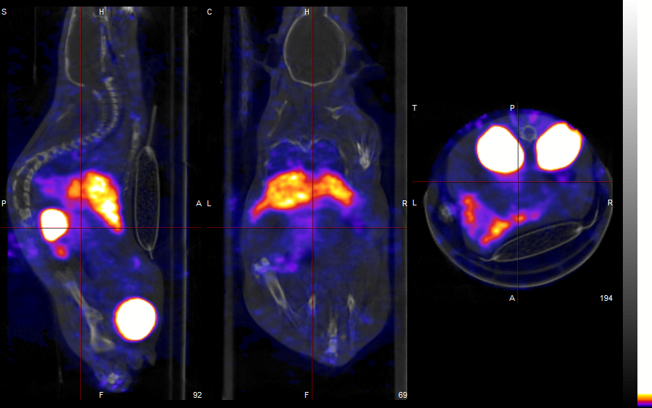
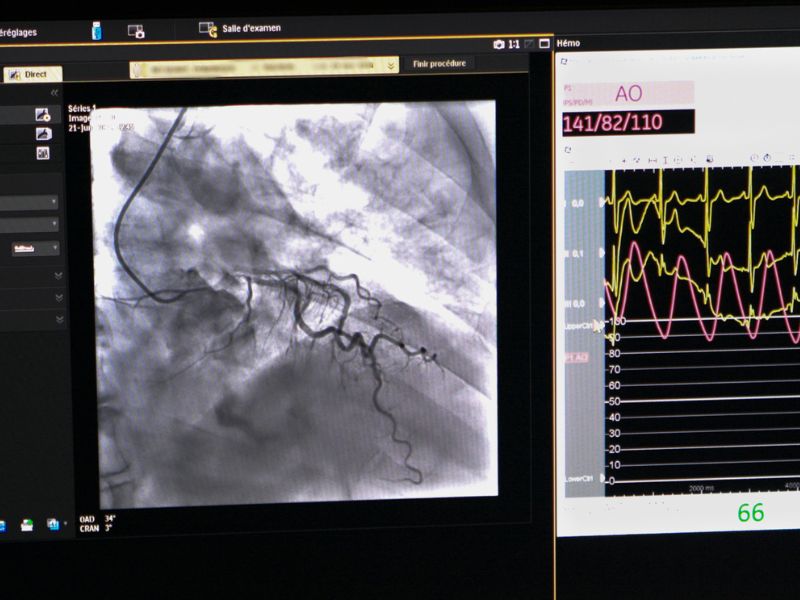
The LRB aims to develop molecules labeled with radioactive atoms for use in nuclear medicine, for diagnostic and therapeutic applications in cardiology, metabolism, oncology, and neurodegenerative diseases.
With integrated biological and clinical components, we cover all steps of a tracer’s development—from the design of the original molecule to its clinical transfer.
All the studies involved in the radiopharmaceuticals development are conducted within our laboratory, including the selection of the biological target for labeling, radiolabeling (radiochemistry), and biological evaluations at multiple levels (cell, organ, and small animal models) before clinical transfer. Then, when a new molecule is transferred, clinical studies (Phase I-IIa, Phase IIb, and Phase III) are carried out at CHUGA in the University Nuclear Medicine Clinic.