- Share
- Share on Facebook
- Share on LinkedIn
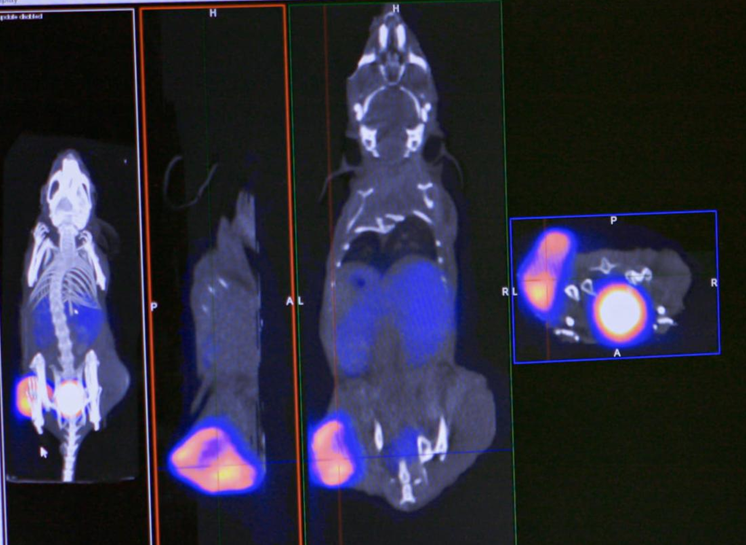
Oncology, alongside Cardiology, is one of the main research themes of the laboratory. Preclinical projects focus on the development of radiopharmaceutical candidates derived from various classes of molecules, such as synthetic molecules (RAFT-RGD), single-domain antibodies or nanobodies (Mesothelin project), and more recently, the development of smart probes that can be activated in vivo (HEALTRIG).
One of the most promising therapeutic approaches in cancer management is Targeted Radionuclide Therapy (TRT), which involves targeting a tumor protein of interest with a specific ligand, labeled with a beta- or alpha-emitting radionuclide. This specific targeting limits irradiation to the ligand binding sites. Since these treatments are relatively intensive, it is essential to first confirm that the target is present on the tumor. This is performed using a diagnostic test (SPECT or PET imaging) with the same ligand, but labeled with a gamma- or positron-emitting radionuclide. The same agent, used either for diagnosis or therapy, is referred to as a theranostic agent.
Current Projects
Mesothelin Project
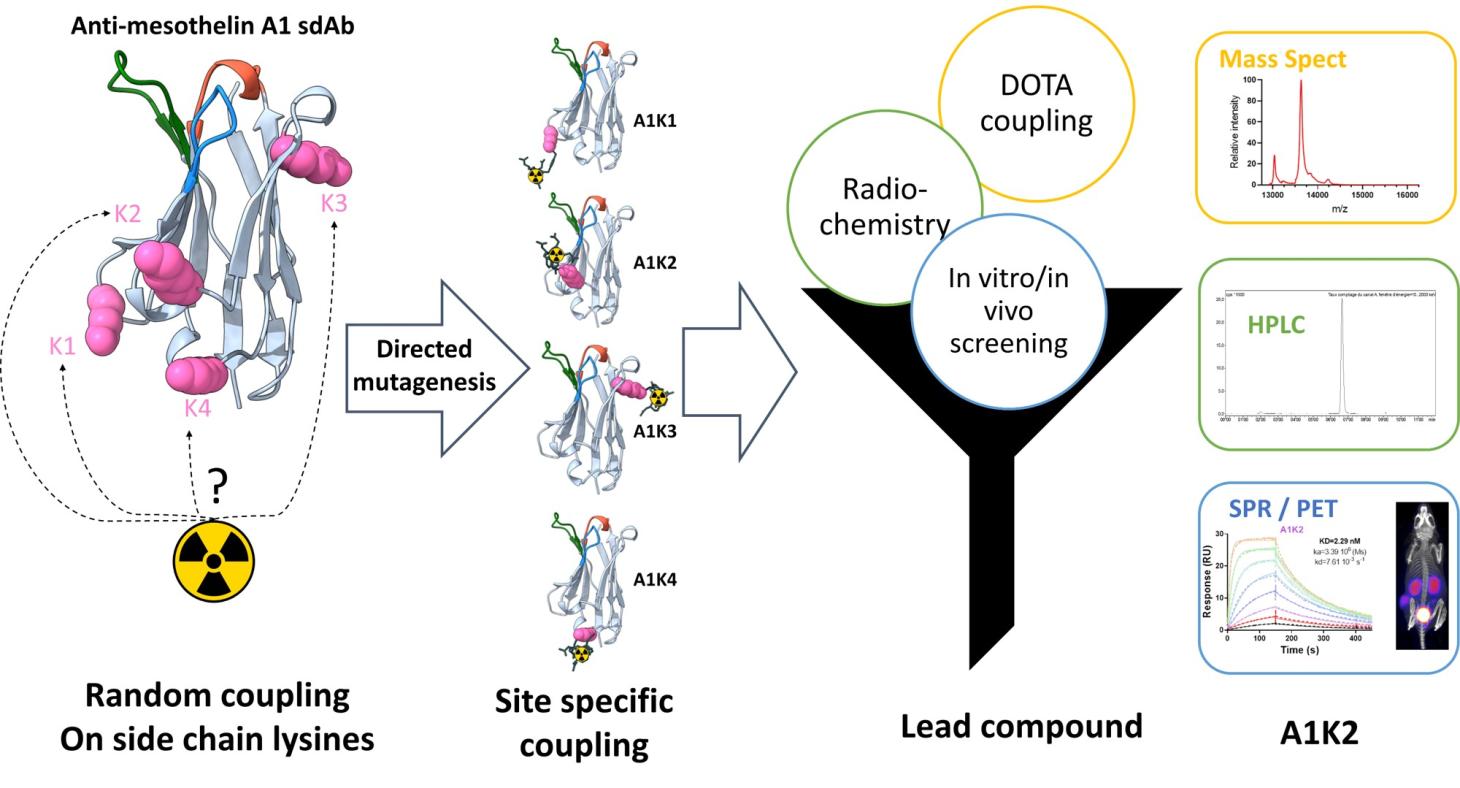
Mesothelin is a protein whose expression in healthy tissues is limited to mesothelial cells lining the pericardium, peritoneum, and pleura. The biological function of mesothelin remains unknown, and no detectable abnormalities have been observed in mesothelin-deficient mice.
However, mesothelin is overexpressed in several human malignant tumors: 100% of epithelial mesotheliomas, the majority of pancreatic and ovarian adenocarcinomas, more than 50% of lung adenocarcinomas, and 34–67% of triple-negative breast cancers. It appears to be involved in tumor aggressiveness, as its expression correlates with a poorer prognosis. The limited expression of mesothelin in healthy tissues and its overexpression in multiple aggressive cancers make it an attractive candidate for the development of new therapies.
As a result, targeted therapies against mesothelin are currently under clinical investigation. In collaboration with the CRCM (Cancer Research Center of Marseille, CNRS UMR 7258, Inserm U1068), we have developed a nanobody labeled with Tc-99m (99mTc-A1) for phenotypic imaging of mesothelin-expressing tumors (MSLN). We then engineered protein derivatives of A1 to create a theranostic agent capable of both diagnostic imaging (PET) and targeted radionuclide therapy.
HEALTRIG Project
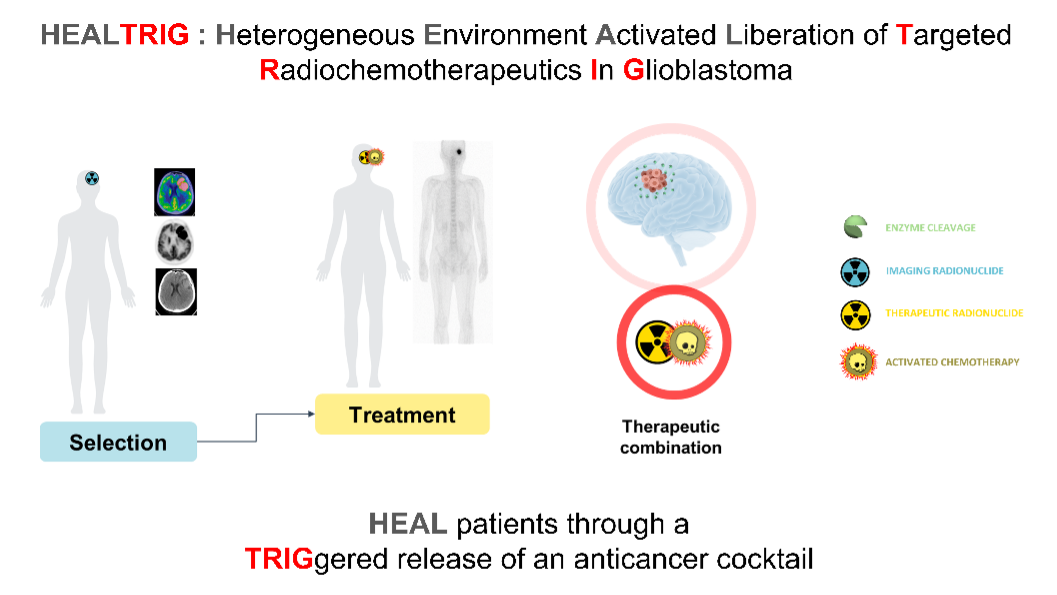
Glioblastoma is the most common and aggressive primary malignant brain tumor. Its poor prognosis highlights the urgent need for more effective, targeted therapeutic approaches. Among emerging strategies, targeting the tumor microenvironment has gained significant attention as a promising approach for innovation.
The HEALTRIG project (Heterogeneous Environment Activated Liberation of Targeted Radiochemotherapeutics in Glioblastoma) aims to develop a theranostic strategy for glioblastoma using programmable molecular systems that respond to β-glucuronidase, an extracellular enzyme specifically present in the tumor microenvironment.
These compounds/systems are co-developed with the OrgaSynth team (“Systèmes moléculaires programmés”) at the IC2MP (Institut de Chimie des Milieux et Matériaux de Poitiers – Institute of Chemistry of Environments and Materials of Poitiers), which specializes in the design of programmable molecular systems for selective drug delivery, stimulus-responsive compounds, volatolomics (the study of volatile biomarkers), chemical biology, and in vivo chemistry.
When activated by β-glucuronidase, the diagnostic version of the molecule releases an imaging radionuclide to identify patients who may benefit from treatment. The therapeutic version delivers both a potent chemotherapeutic agent, activated locally within the tumor, and a therapeutic radionuclide.
A key advantage of the HEALTRIG approach lies in its innovative mechanism of action. Unlike conventional strategies that rely on a single molecular target/specific biomarker, these programmable systems are designed to trigger selective activation directly within the tumor environment. This approach is particularly well-suited to oncology, where tumor heterogeneity often limits the effectiveness of traditional therapies.
By enabling localized drug release across diverse tumor types, without depending on a single specific biomarker, these molecules hold potential for broader application beyond glioblastoma, opening new possibilities for the treatment of various tumor pathologies.
Once proof of concept is established, this personalized theranostic strategy is expected to reduce off-target toxicity and minimize the dose-limiting side effects often encountered with conventional, non-targeted chemotherapy.
Several complementary projects are now emerging around HEALTRIG, exploring how the physical properties of therapeutic radionuclides can be used to trigger the controlled release of chemotherapeutic agents, or to enhance therapeutic efficacy through combination strategies.
RAFT-RGD Project
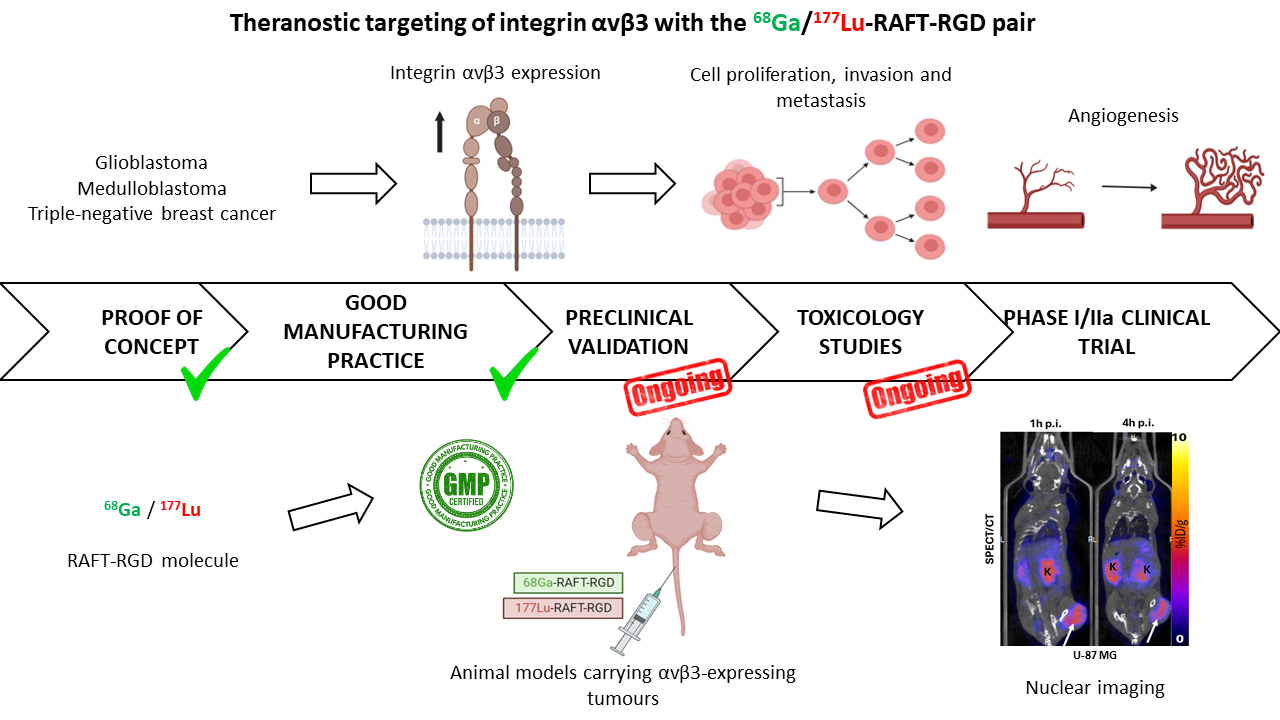
Solid tumors such as glioblastoma (GBM), medulloblastoma (MB), and triple-negative breast cancer (TNBC) represent major challenges in oncology. Their aggressiveness, resistance to conventional treatments, and often poor prognosis necessitate the development of innovative therapeutic strategies. Integrin αvβ3, an adhesion molecule involved in angiogenesis and tumor progression, is highly expressed in these cancers, making it a strategic target.
A promising theranostic agent targeting integrin αvβ3 is under investigation in our laboratory. It has been radiolabeled with Ga-68 for diagnostic purposes and with Lu-177 for therapy. Preclinical studies have demonstrated the theranostic potential of this agent. A clinical study involving patients with either metastatic breast cancer or glioblastoma will be conducted at the Grenoble-Alpes University Hospital (CHUGA) in a Phase I/IIa clinical trial. The first-in-human injection is scheduled for June 2026.
Contact
Principal Investigators
Mesotheline:
Alexis Broisat
alexis.broisat inserm.fr (alexis[dot]broisat[at]inserm[dot]fr)
inserm.fr (alexis[dot]broisat[at]inserm[dot]fr)
Tel +33 (0)4 76 63 71 02
HEALTRIG:
Florian Raes
florian.raes inserm.fr (florian[dot]raes[at]inserm[dot]fr)
inserm.fr (florian[dot]raes[at]inserm[dot]fr)
RAFT-RGD:
Catherine Ghezzi
catherine.ghezzi univ-grenoble-alpes.fr (catherine[dot]ghezzi[at]univ-grenoble-alpes[dot]fr)
univ-grenoble-alpes.fr (catherine[dot]ghezzi[at]univ-grenoble-alpes[dot]fr)
Tel +33 (0)4 76 63 74 76
- Share
- Share on Facebook
- Share on LinkedIn