- Share
- Share on Facebook
- Share on LinkedIn

Background
With over 537 million people affected worldwide, and projections rising to 783 million by 2045, diabetes has become a major global public health challenge [GBD Diabetes Coll, 2023]. It is a chronic condition diagnosed by a fasting blood glucose level ≥ 1.26 g/L that occurs when the pancreas no longer produces enough insulin and/or when the body no longer responds correctly to insulin/becomes resistant to the insulin it produces (insulin resistance or IR).
There are two main types of diabetes:
- Type 1 diabetes (T1D) is an autoimmune disease typically occurring in children or young adults, leading to the progressive destruction of pancreatic β cells.
- Type 2 diabetes (T2D) occurs mostly in adults, with over 80% of cases associated with overweight or obesity.
In addition to IR, obesity is also associated with a chronic inflammatory response, characterized by abnormal cytokine production from adipose tissue and activation of pro-inflammatory signaling pathways. This association is not incidental, numerous animal models have shown that this inflammatory response plays a causal role in the development of obesity-related comorbidities: including IR, T2D and atherosclerosis.
Projects
The metabolic diseases research axis includes three major projects:
Molecular Imaging of Pancreatic β Cell
Development of new radiotracers to assess functional pancreatic β-cell mass by nuclear imaging
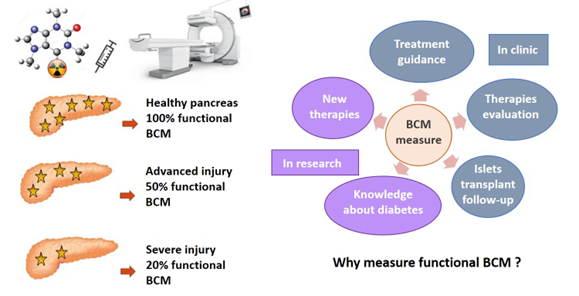
The key function of pancreatic β cells is to secrete insulin in response to rising blood glucose levels. These cells are clustered within the islets of Langerhans, which make up only 1 to 2% of the total pancreatic mass and are widely dispersed throughout the exocrine tissue.
A reduction in pancreatic β-cell mass (BCM), whether anatomical or functional, is a common feature of both T1D and T2D. The ability to assess BCM in vivo would enable clinicians to evaluate and monitor the anatomical and functional status of the patient’s pancreas and ajust treatment accordingly. Furthermore, with advances in β-cell transplantation for the treatment of T1D, there is a growing need for imaging tools that can monitor graft function.
In research, such imaging tools would also support the evaluation of new therapeutic strategies. Although various techniques have been explored, including MRI, PET or SPECT, none has yet reached clinical application.
This project aims to develop a new β-cell specific radiotracer suitable for routine use in nuclear medicine, capable to quantify/measure functional mass in vivo via nuclear imaging.
Imaging Inflammation in MASLD
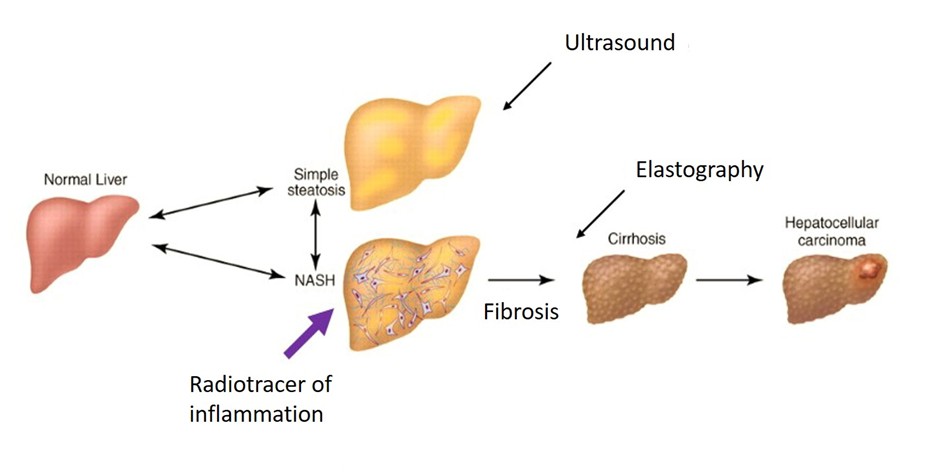
Metabolic dysfunction-Associated Steatotic Liver Disease (MASLD) is usually asymptomatic, at least until advanced/later stages. As a result, the disease often progresses silently, leading ultimately to cirrhosis and/or hepatocellular carcinoma. It is characterized by the excessive accumulation of lipids in the liver and includes a wide spectrum of liver damage, ranging from simple steatosis to steatohepatitis (MASH), which affects about/approximately one third of MASLD patients.
Unlike simple steatosis, MASH is associated with a poorer prognosis in the long term/run, progressive fibrosis and an increased cardiovascular risk. MASLD is a growing public health concern, strongly linked to obesity. Despite its high and rising prevalence, there is currently no effective non-invasive method to diagnose early-stage disease, and no approved treatment exists.
This project aims to develop and validate a non-invasive nuclear imaging method/technique to detect early markers of the disease/MASLD, in particular inflammation.
Highlights
Nachit M, Montemagno C, Clerc R, Ahmadi M, Briand F, Bacot S, Devoogdt N, Serdjebi C, Ghezzi C, Sulpice T, Broisat A, Leclercq IA, Perret P. Molecular imaging of liver inflammation using an anti-VCAM-1 nanobody. Nat Commun 14(1):1062, 2023. https://www.nature.com/articles/s41467-023-36776-7
A license agreement was granted by UGA (Université Grenoble Alpes) and VUB (Vrije Universiteit Brussel) to develop the application of the cAbVCAM1-5 nanobody in MASLD.
Molecular Imaging of Cellular Senescence
Cellular senescence is a permanent state of cell cycle arrest, associated with metabolic dysregulation, macromolecular damage, and the acquisition of a pro-inflammatory secretory phenotype. These alterations can lead to a loss of tissue function. Senescent cells contribute to/play a role in many chronic diseases, including metabolic liver diseases and heart failure.
Senotherapies, which are treatments designed to target senescent cells specifically, offer an innovative therapeutic approach with a wide range of potential applications for age-related and chronic conditions. However, evaluating these agents and, in the longer term, monitoring treatment response remain challenging due to the lack of reliable methods to detect and measure senescent cell burden in vivo.
In response to this challenge, a project at the LRB aims to develop a molecular imaging agent capable of visualizing senescent cell burden non-invasively.
Contact
Principal Investigators
Beta Cell:
Pascale Perret
pascale.perret univ-grenoble-alpes.fr (pascale[dot]perret[at]univ-grenoble-alpes[dot]fr)
univ-grenoble-alpes.fr (pascale[dot]perret[at]univ-grenoble-alpes[dot]fr)
Tel +33 (0)4 76 63 71 02
MASLD:
Pascale Perret
and
Alexis Broisat
alexis.broisat inserm.fr (alexis[dot]broisat[at]inserm[dot]fr)
inserm.fr (alexis[dot]broisat[at]inserm[dot]fr)
Senescence:
Jakub Toczek
jakub.toczek inserm.fr (jakub[dot]toczek[at]inserm[dot]fr)
inserm.fr (jakub[dot]toczek[at]inserm[dot]fr)
- Share
- Share on Facebook
- Share on LinkedIn