- Share
- Share on Facebook
- Share on LinkedIn

Background
Radiochemistry is central to many research projects in preclinical imaging and nuclear medicine. Positioned at the crossroads of chemistry, nuclear physics, and medicine, it involves labeling a targeting molecule (also called a ligand), capable of selectively binding to specific biological structures, with a radionuclide. This approach enables the development of radiotracers used for diagnosis (SPECT or PET imaging), as well as for targeted radionuclide therapy.
The core focus of radiochemistry is the synthesis and analysis of radiotracers tailored to the biological or therapeutic objectives of each study. This includes designing efficient radiosynthesis pathways and developing reliable analytical methods for each compound.
Radiochemistry at the LRB
Radiochemistry at the LRB focuses on the development, production, and analysis of novel radiotracers that are essential to the laboratory’s internal research projects, scientific collaborations, and the services provided through the GAIA platform.

The radiochemistry lab is organized into two complementary areas:
- SPECT radiochemistry lab: This facility is dedicated to radiotracers labeled with gamma-emitting radionuclides (e.g., 99mTc, 125I, 123I) or beta-emitting radionuclides (e.g., 177Lu, 90Y).
- PET radiochemistry lab: Installed more recently, in 2020, this area is specifically designed and equipped for the preparation of positron-emitting radiotracers (e.g., 68Ga, 64Cu).
These facilities support innovative and safe research in full compliance with regulatory standards and are structured to facilitate the transfer of technologies to clinical practice and industry.
From Radiolabeling to Injection: Key Steps
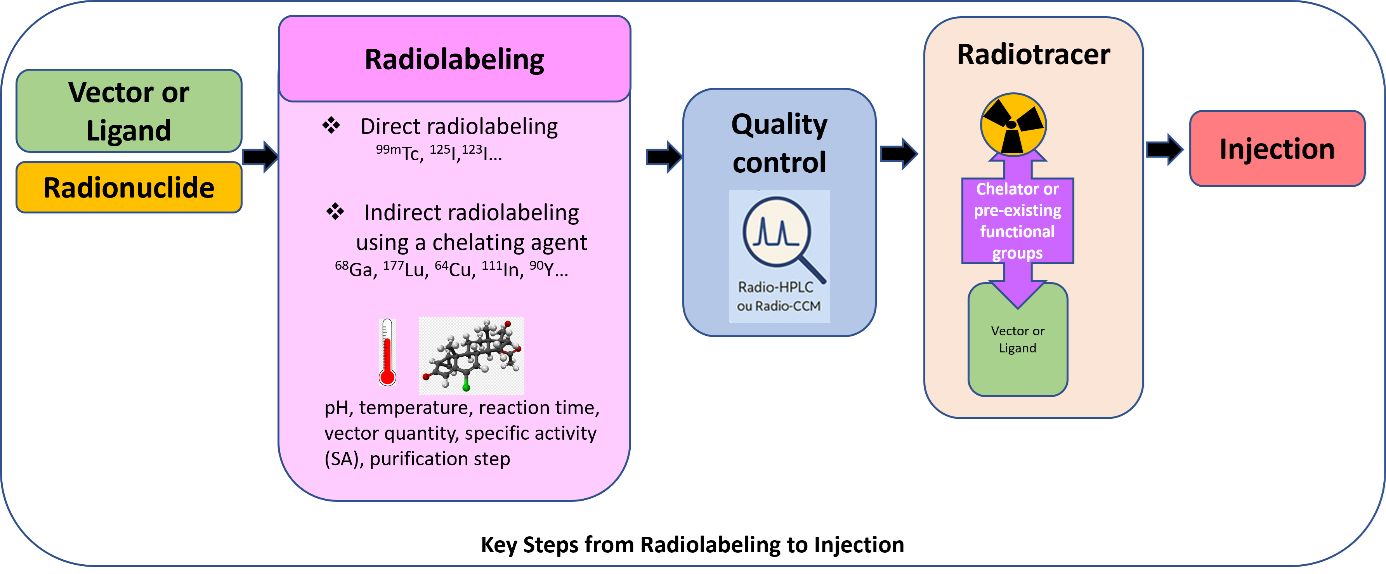
-
Selection of Targeting Molecules and Radionuclides
A wide range of biologically or therapeutically relevant molecules (vectors or ligands), from diverse chemical families can be radiolabeled. These include biomolecules (e.g., peptides, proteins, antibodies, nanobodies, carbohydrates), synthetic compounds, cells (e.g., whole cells, blood cells, bacteria), or oligonucleotides (such as siRNAs).
Radionuclides are chosen based on the intended biological application. For diagnostic purposes, gamma emitters (e.g., 99mTc, 125I, 123I, 111In) are used for SPECT imaging, while positron emitters (e.g., 68Ga, 64Cu) are used for PET imaging. For targeted radionuclide therapy, other radionuclides are used such as beta- emitters (177Lu, 90Y, or 161Tb) and in the near future, alpha emitters like 225Ac (authorization obtained). -
Radiolabeling
The process begins with selecting an appropriate radiolabeling strategy: either direct labeling or indirect labeling using a chelator, which must first be conjugated to the targeting molecule. Radiolabeling conditions are then optimized, based on the complexation conditions that are tailored the properties of both the ligand and the radionuclide, to ensure high radiochemical purity (RCP) and optimal specific activity (SA), all within timeframes compatible with the radionuclide's half-life.
Other radiosynthesis parameters such as pH, temperature, incubation time, and activity-to-molecule ratio (specific activity) are also optimized. If needed, purification steps are included to improve the final quality of the radiotracer. The final formulation must be injectable, meeting requirements for solubility, excipient or solvent tolerance, and volumetric activity appropriate for the selected animal models. -
Quality Control
Throughout the process, validated analytical techniques, such as radio-HPLC and radio-TLC, are employed to ensure the quality, purity, and reproducibility of the radiotracers.
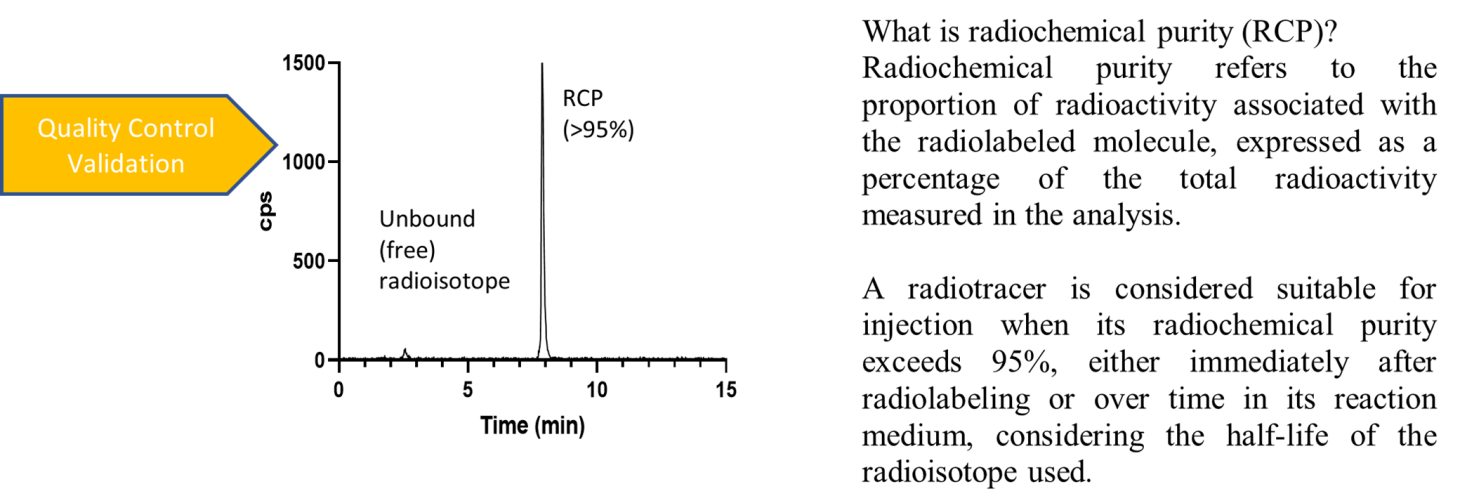
Assessment of In Vitro and Ex Vivo Stability
Radiotracer stability can be evaluated in vitro using culture media or specific buffers/solvents relevant to downstream experiments, as well as in whole blood, plasma or serum from animals (mice, rats, rabbits) or from human donors (through agreement with the French Blood Establishment). Ex vivo stability testing can also be performed by analyzing blood samples collected via cardiac puncture in animals.
Toward Clinical Translation
For translational applications, radiosynthesis parameters are adjusted to comply with pharmaceutical standards, ensuring that radiotracers meet the regulatory requirements for clinical-grade production.
Contact
Sandrine Bacot
sandrine.bacot univ-grenoble-alpes.fr (sandrine[dot]bacot[at]univ-grenoble-alpes[dot]fr)
univ-grenoble-alpes.fr (sandrine[dot]bacot[at]univ-grenoble-alpes[dot]fr)
- Share
- Share on Facebook
- Share on LinkedIn